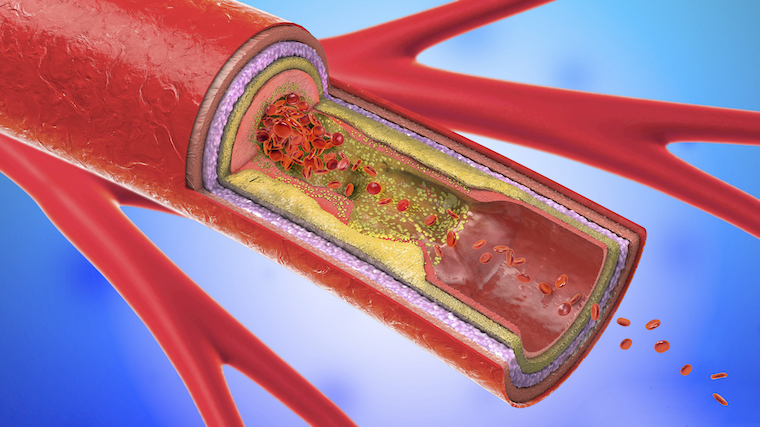
Coronary artery disease is caused by the build up of fatty plaques in the vessels that supply blood to the heart.
UVA Health researchers probing the causes of coronary artery disease – the nation’s No. 1 killer – have identified critical biological processes that can go wrong inside the linings of our blood vessels and contribute to the dangerous plaque buildup responsible for the disease.
The discovery provides new targets for scientists seeking to develop better ways to treat and prevent coronary artery disease (CAD), a condition responsible for 1 in 4 deaths in the United States. More than 600,000 Americans die from the disease every year, and more than 17 million people die from it worldwide.
“Smooth muscle cells that make up the bulk of our blood vessels play important roles in coronary artery disease. They undergo pathological transformations as the disease develops inside our arteries,” said researcher Mete Civelek, PhD, of the University of Virginia School of Medicine’s Center for Public Health Genomics and the Department of Biomedical Engineering. “Our results point to a previously under-appreciated role for metabolic pathways during this pathological transformation.”
Coronary Artery Disease: A Strange Mystery
Civelek and his team wanted to unravel a longstanding mystery about the behavior of smooth muscle cells during plaque formation. These cells, which line our blood vessels, are integral in protecting the body during plaque formation – they build stabilizing caps over the plaques that prevent the lesions from breaking loose and causing strokes. But sometimes these beneficial smooth muscle cells take off their white hats and put on black ones. Instead of helping to stabilize the plaques, they begin to accelerate the plaque development and spur the progression of CAD, scientists believe.
Civelek’s new discovery helps explain why. Noah Perry, a PhD student on Civelek’s team, analyzed smooth muscle cells collected from 151 heart transplant donors and used a sophisticated approach to identify genes responsible for the smooth muscle cells’ behavior.
After initially identifying 86 groups of genes, the researchers focused in on 18 groups that could explain the mysterious behavior. Their analysis suggested that the smooth muscle cells’ shift to the dark side might stem from problems with how the cells use nitrogen and glycogen. (Glycogen is how the body stores the sugar glucose.)
Further, the researchers identified a particular sugar, mannose, that may be contributing to those problems – potentially even triggering them. But determining that, the scientists say, will require more research.
“The metabolic shift in the cells as they transition to a disease state can point to points of intervention and therapy,” said Perry, of UVA’s Department of Biomedical Engineering and the lead author of the study.
By better understanding what triggers the smooth muscle cells to become harmful, Civelek says, doctors may be able to develop ways to prevent that from happening. That could open the door to new ways to treat and prevent coronary artery disease.
“Coronary artery disease is still the leading cause of death worldwide,” Civelek said. “Although cholesterol-lowering therapies and blood pressure control have been very effective tools to prevent deaths from heart attacks, we still need more targets to reduce the suffering of patients and their families from this devastating disease.”
Findings Published
The researchers have published their findings in the scientific journal Circulation: Genomic and Precision Medicine. The research team consisted of Perry, Diana Albarracin, Redouane Aherrahrou and Civelek. The researchers have no financial interest in the work.
The coronary artery disease research was supported by the National Institutes of Health, grants T32 HL007284, F31 HL165772 and R01 HL166428; American Heart Association Postdoctoral Fellowship 18POST33990046; Transformational Project Award 19TPA34910021; and International Network of Excellence Award 22CVD04 from Foundation Leducq.
UVA’s Department of Biomedical Engineering is a joint program of the School of Medicine and the School of Engineering.
To keep up with the latest medical research news from UVA, subscribe to the Making of Medicine blog.